Each person from the UC Irvine team that we've interacted with has been nothing but positive, flexible, and understanding of our common goals.
Our Process
Contact our Business Development Specialist
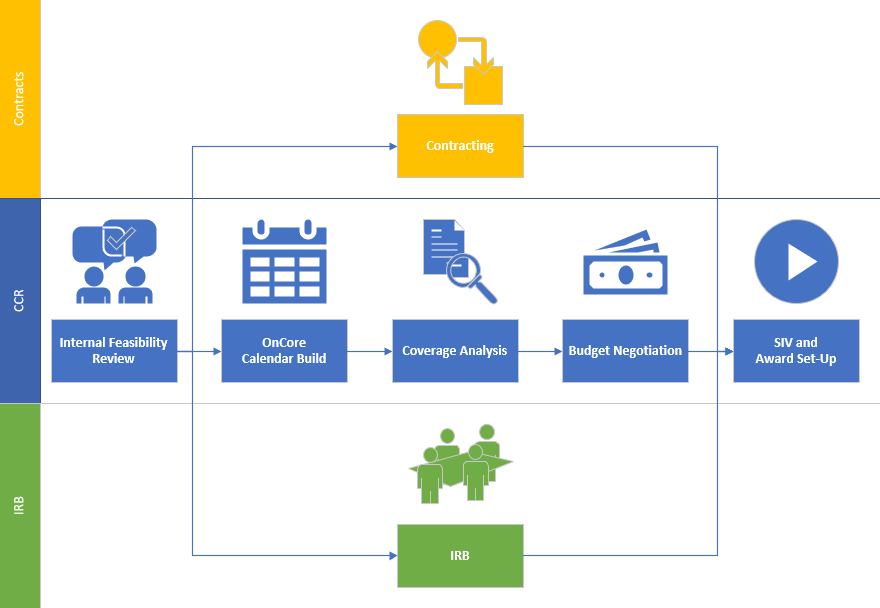
At UCI Center for Clinical Research (CCR), we are fiercely dedicated to growing our clinical trial portfolio and ensuring that our patients and community have access to the latest and most innovative treatment options as part of their care. To achieve this, we have developed high-performance teams that are committed to activating all clinical trials within 120 days.
Our process begins when a sponsor contacts our Business Development (BD) team about a potential opportunity. Our BD Specialists are experts in introducing sponsors to our process, executing confidentiality disclosure agreements (CDAs), and identifying a Principal Investigator (PI) at UCI.
Once the sponsor submits all the essential documents to our team - including the study protocol, the informed consent form, the study budget template, and the initial agreement - our clock officially starts. The study then goes to our weekly Internal Feasibility Committee (IFC), and once it's approved, it's time to "Start Your Engines!"